For example, look at a simple chemical reaction: when we heat limestone, which is made of calcium carbonate (CaCO3), it liberates a gas, carbon dioxide (CO2), and we obtain a solid residue, calcium oxide (CaO):

Is this chemical reaction adjusted? Let's analyse it:
- CaCO3(s) → CaO(s) + CO2(g)
On the left there is an atom of carbon, and on the right there is one, too.
On the left there are three atoms of oxygen, and on the right there are three, too.
Therefore, it is adjusted: for every molecule of calcium carbonate (CaCO3) we will obtain 1 molecule of calcium oxide (CaO) and 1 molecule of carbon dioxide (CO2).
Now look at the chemical reaction that takes place when we burn hydrogen and obtain water.
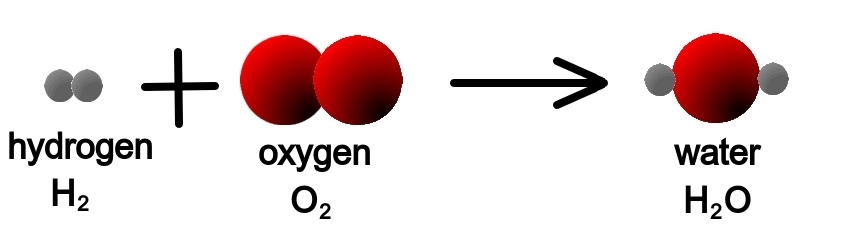
- H2(g) + O2(g) → H20(g)
On the right we also have two atoms of H, so that is all right.
On the left we have a molecule of O (two atoms of O form a molecule of O2 gas)
However, on the right we have only one atom of O in the molecule of gaseous water.
Therefore, this chemical reaction is not adjusted.
To adjust it, we need to put TWO molecules of water on the right,
so that we have two atoms of oxygen:
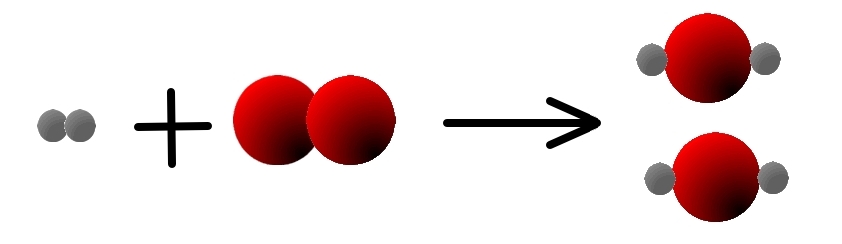
- H2(g) + O2(g) → 2 H20(g)
so we need to have 2 molecules of H2 on the left:
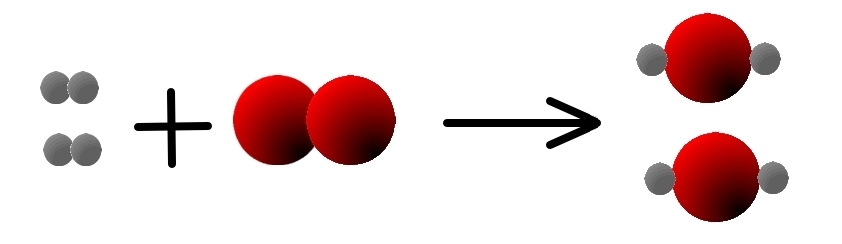
- 2 H2(g) + O2(g) → 2 H20(g)
and we have 2 atoms of O on the left, and 2 on the right,
so, this chemical reaction IS balanced.
Look at a more difficult example: when we burn methane gas we obtain carbon dioxide and water:
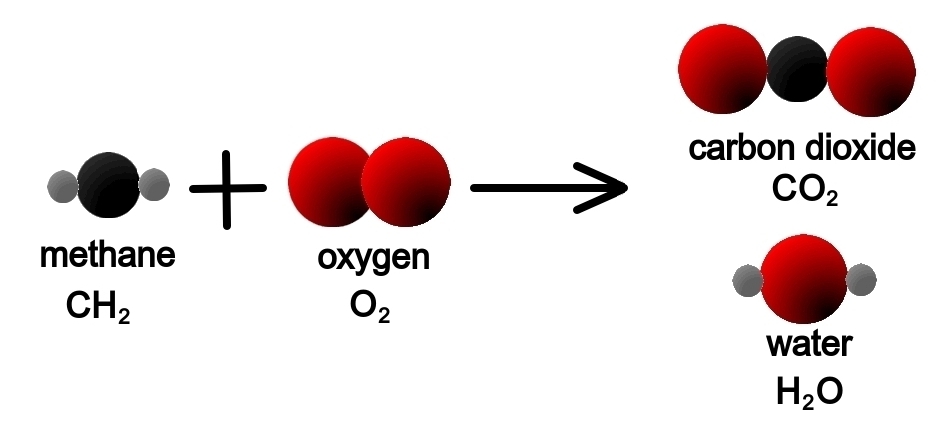
- CH2(g) + O2(g) → C02(g) + H20(g)
On the left we have one atom of carbon in the molecule of methane, and on the right we have one, too, in the molecule of carbon dioxide.
On the left we have two atoms of hydrogen in the molecule of methane, and on the right we have two, too, in the molecule of water.
However, on the left we have two atoms of oxygen in the molecule of oxygen, while on the right we have three: two in the molecule of carbon dioxide, and another in the molecule of water.
If we want to add more oxygen on the left, we need to add more methane, too:
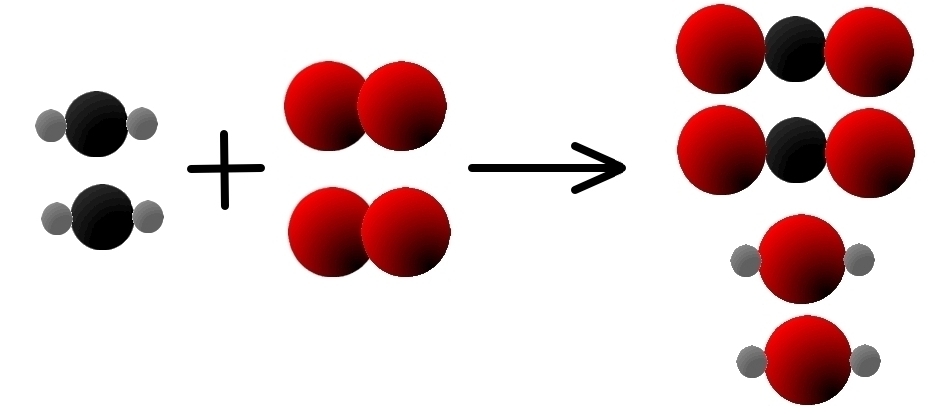
- 2 CH2(g) + 2 O2(g) → C02(g) + H20(g)
We will also have 4 atoms of hydrogen in the methane on the left, and we will get four in the two molecules of water on the right, so the hydrogen is balanced, too.
However, the oxygen is not balanced yet: we have 4 atoms of oxygen on the left and 6 on the right, so we need to add another molecule of oxygen on the left:
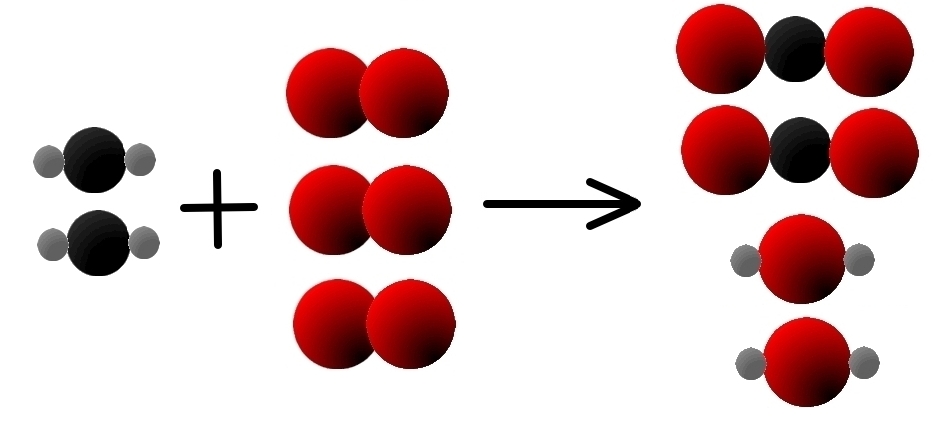
- 2 CH2(g) + 3 O2(g) → 2 C02(g) + 2 H20(g)
Now, balance this chemical reaction in your notebook. It takes place when iron, a solid, combines with oxygen, a gas, and gets rusted (the iron becomes iron oxide):
- Fe(s) + O2(g) → Fe3O2(s)
[
There is 1 atom of Fe on the left, but 3 on the right, so we need 3 atoms of Fe:
- 3 Fe + O2 → Fe3O2
Now, balance another chemical reaction in your notebook. It takes place when water and the metal sodium react and form sodium hydroxide (NaOH) and hydrogen gas (H2):
- Na(s) + H2O(l) → NaOH(s) + H2(g)
[
There is 1 atom of Na on the left and one on the right, so the Na is balanced.
There is 1 atom of O on the left and one on the right, so the O is balanced, too.
However, there are 2 atoms of H on the left and 3 on the right.
We add more Na and more water; now we have 2 atoms of Na, so we get two molecules of NaOH:
- 2 Na + 2 H2O → 2 NaOH + H2