




- The wheel will be stronger if the nails are evenly spaced, that is, regularly distributed.
- The atom's electrons will be more stable if the outer layer has all the available spaces filled with electrons.
Electron configuration
For some atoms it is easier to combine with other atoms, because they can easily lose or gain electrons,
but why are the electrons in some atoms more stable than in others?
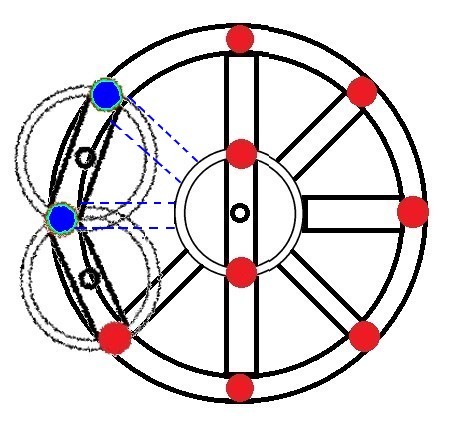
We can compare the stability of the electrons in an atom's orbit with the strength of a wooden wheel:
 |
Primitive wheels were very heavy, but not very strong. |
 |
Later, wheels became lighter and stronger (and easier to make and repair), thanks to radia. |
 |
The more radia a wheel had, the lighter and stronger it was. |
 |
In an atom, as in this wheel, there is usually more than one layer of electrons (only hydrogen and helium have a single layer). |
 |
We can imagine electrons as nails holding together the radia to the wheel's axis and circumference:
|
Remember that layers do not fill with atoms consecutively, but following the Aufbau principle
(also known as "Moeller's rubric"):
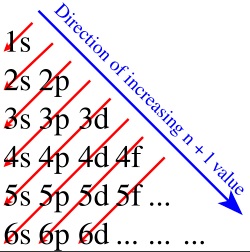
Study the electron configuration of these atoms to understand it better:
|
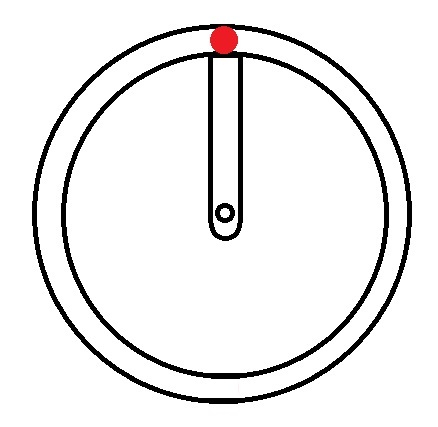
Hydrogen → 1 electron → 1s1 |
 |
| |
|
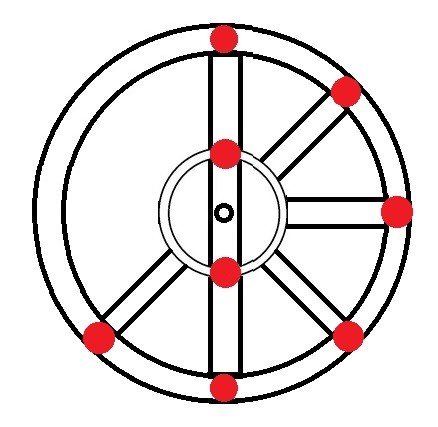
Oxygen → 8 electrons → 1s2 2s2 2p4 |
 |
|
How do atoms combine to form molecules?
|
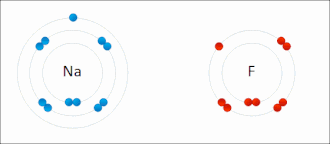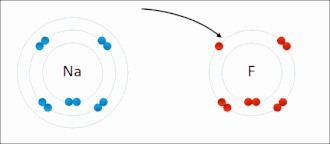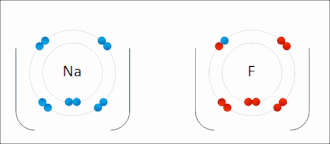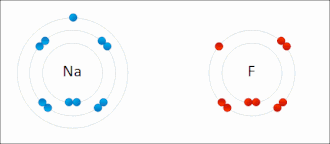
Salt (sodium chlorine) → ionic bonding |
 |
|
|
Water (H2O) → covalent bonding |
 |
|
(Metallic bonding is very different)
Why do some atoms not react with others?
|
Helium → 2 electrons → 1s2 |
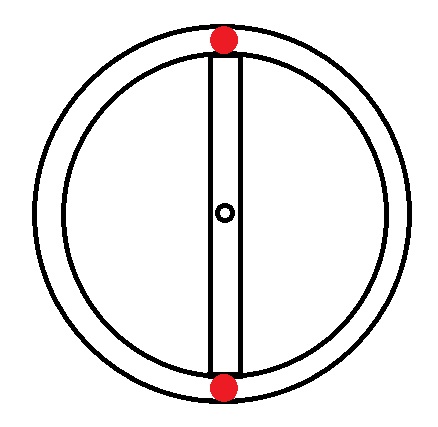 |
| |
|
Ne → 10 electrons → 1s2 2s2 2p6 |
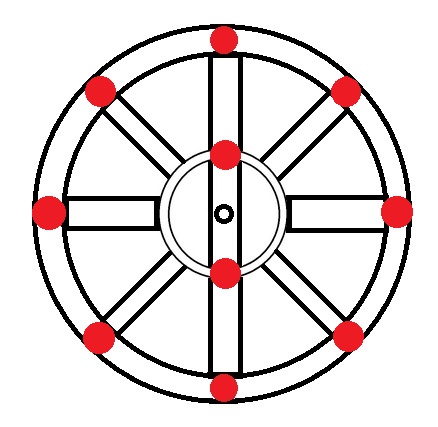 |
| |
|
Carbon → 6 electrons → 1s2 2s2 2p2 |
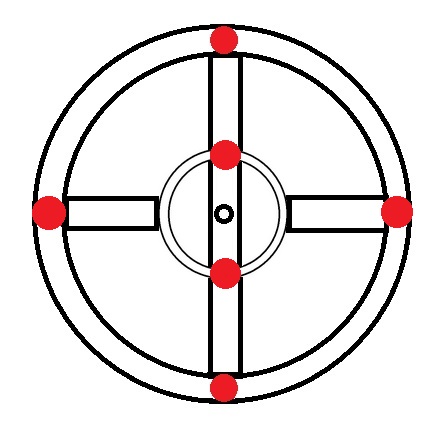 |
|
|
|
Silicon → 14 electrons → 1s2 2s2 2p6 3s2 3p2 |
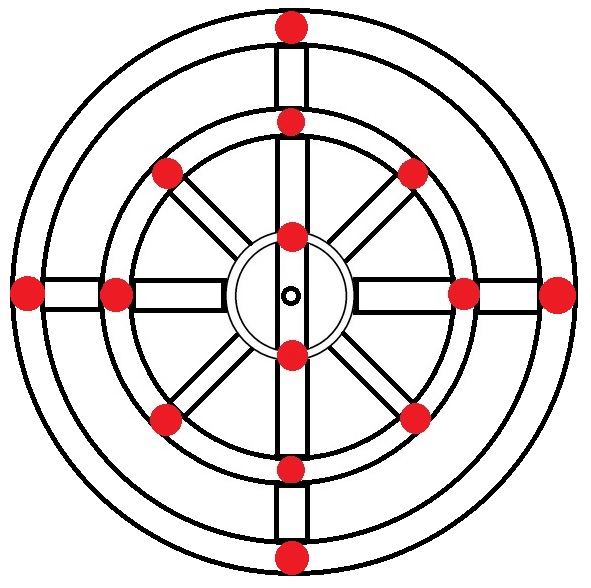 |
|